- Preparation of Acrylate Emulsions in the CuBr2/PMDETA Catalytic System by the ARGET–ATRP Method and Kinetic Studies
Xingbing Yang*, ***, ****,†
 , Xinye Wang**,†
, Xinye Wang**,†  , Wenyu Yan*, Yi Xie*, Xinyue Xu*, Yanxue Li*, and Gang Chen*
, Wenyu Yan*, Yi Xie*, Xinyue Xu*, Yanxue Li*, and Gang Chen**College of Chemistry and Chemical Engineering, Sichuan University of Arts and Science
**Guangxi Key Laboratory of Electrochemical and Magnetochemical Function Materials,
College of Chemistry and Bioengineering, Guilin University of Technology
***Key Laboratory of Low-cost Rural Environmental Treatment Technology, Sichuan University of Arts and Science
****Special Polymer Materials for Automobile Key Laboratory of Sichuan Province, Sichuan University of Arts and Science- ARGET-ATRP법에 의한 CuBr2/PMDETA 촉매 시스템에서의 아크릴레이트 에멀전 제조 및 속도론 연구
Reproduction, stored in a retrieval system, or transmitted in any form of any part of this publication is permitted only by written permission from the Polymer Society of Korea.
The activators regenerated by electron transfer–atom transfer radical polymerization (ARGET–ATRP) technique was utilized in the emulsion copolymerization of 2-ethylhexyl acrylate (2-EHA), ethyl acrylate (EA) and methyl methacrylate (MMA) as the copolymer raw material, and ethyl 2-bromoisobutyrate (EBIB) was used as the initiator in a pentamethyldivinyltriamine (PMDETA)/copper bromide-catalyzed system for emulsion copolymerization. The effects of the reaction temperature, reducing agent ascorbic acid (AA) dosage and ligand concentration on the copolymerization kinetics were investigated. The synthesized polymers were characterized by proton nuclear magnetic resonance (1H NMR) spectroscopy and infrared (IR) spectroscopy. The results showed that the copolymerization monomers added to the reaction were successfully initiated by the initiator and polymerized into terpolymers, the conversion rate of the monomers gradually increased with increasing amounts of ligand and reductant, and the conversion rate of the monomers involved in the copolymerization reaction and the apparent reaction constants gradually increased with increasing reaction temperature. The apparent activation energy of the copolymerization reaction, Ea, was calculated to be 80.43 kJ·mol-1.
In this study, the activators regenerated by electron transfer–atom transfer radical polymerization (ARGET-ATRP) method was employed under the catalytic system of CuBr2/pentamethyldivinyltriamine (PMDETA) to successfully carry out the emulsion polymerization of 2-ethylhexyl acrylate (2-EHA), methyl methacrylate (MMA), and ethyl acrylate (EA). The effects of the amount of ligand, the amount of reducing agent, and the reaction temperature on the copolymerization reaction kinetics were also investigated.
Keywords: activators regenerated by electron transfer–atom transfer radical polymerization, kinetics, emulsion copolymerization, apparent activation energy, apparent polymerization rate constant
Special Polymer Materials for Automobile Key Laboratory of Sichuan Province at Sichuan University of Arts and Science (TZGC2024ZA-04, TZGC2023ZB-04).
Atom transfer radical polymerization (ATRP), as the core technique for controlled/activated radical polymerization, has demonstrated theoretical and practical importance in the precise synthesis of polymeric materials. Conventional ATRP systems achieve reversible activation of radicals through low-valent transition metal catalysts, but their high sensitivity to oxidative environments severely limits their industrial application. Reverse ATRP (RATRP) considerably reduces the dependence of the reaction on the anoxic environment through a unique reaction mechanism, in which high-valent metal complexes are used as the initial catalysts, and active catalytic species are generated in situ via the redox action of the initiator, laying the foundation for a breakthrough in the practicability of the system. The ATRP of activators regenerated by electron transfer (ARGET–ATRP) further optimizes the catalytic system by reducing the amount of transition metals to the ppm level, and the residual catalysts after the reaction do not require additional treatment, which greatly simplifies the production process. Given the special requirements for cost control and water compatibility in industrial applications, this study selects PMDETA to replace the conventional ligand TPMA. This decision is mainly based on two considerations: First, the cost advantage. The market price of PMDETA is only one-third that of TPMA. The second is water solubility compatibility. Its higher solubility in the aqueous emulsion system can reduce the decrease in catalytic efficiency caused by ligand agglomeration. In research on the optimization of the ARGET–ATRP system, Dhar et al. demonstrated that ARGET-ATRP exhibits one-stage kinetics at different temperatures when methyl methacrylate is used as the monomer, which verifies the controllability of the polymerization. Yang et al.7 used this method to synthesize novel block copolymers and prepare dispersants modified with amine groups, which expands the boundaries of the applications of functional materials. Kwak et al. used a low-valent ligand in synergistic interactions with zero-valent copper at 25 ℃ to simultaneously synthesize a new type of block copolymer at the ppm level, and the residual catalysts did not require additional treatment. Notably, the ratio of catalyst to reducing agent used in AGET–ATRP is 1:1, whereas ARGET–ATRP can be polymerized with less catalyst and more reducing agent.9 Owing to the differences in systems, the amount of catalyst used in ARGET–ATRP varies. As shown in the following three articles,10-12 the ARGET–ATRP polymerization method was adopted. However, owing to the differences in the system and monomer components, the amount of catalyst used varies greatly. In the early stage of the experiment, Kwak et al. used a catalyst ten times lower than that in this paper for copolymerization, but the reaction progress was too slow, with a conversion rate of only 16.70% after 10 hours. Therefore, a catalyst of 0.075 g was selected in the later stage, and the conversion rate significantly improved. In this study, which was based on the method of Simakova et al.11 the molar ratio of the catalyst to the monomer was 9.94×10-4, which was similar to the results reported in the literature. To address the metal catalyst pollution problem, metal-free ATRP systems have become a popular research topic. These systems use organic photocatalysts to regulate free radicals via an oxidation–reduction burst mechanism, which provides a new idea for green polymerization technology. At present, controlled free radical polymerization can synthesize macromolecules with various topological structures, such as blocks, combs and stars, and has become a central means of regulating the physicochemical properties of materials. Since the industrialization of emulsion polymerization technology in the 1930s, batch, semicontinuous and continuous processes have been gradually developed. The batch type is easy to operate and suitable for mass production; the continuous type has become the first choice for large-scale industrialization because of its high efficiency and low cost, but it requires very high equipment precision and process control. When water is used as the dispersion medium, emulsion polymerization completely avoids the volatile organic compound (VOC) pollution caused by the volatilization of organic solvents in traditional polymerization, which is consistent with the concept of green chemistry. The high specific heat capacity of the water medium can buffer the reaction heat and precisely regulate the polymerization rate and product quality. Zhu et al. synthesized organo-attapulgite/polyacrylate nanocomposite latexes by soapless emulsion polymerization, which significantly increased the mechanical strength, water resistance and thermal stability of the films through homogeneous dispersion of the nanoparticles and optimization of the interfacial bonding, providing a new approach to high-performance coatings and composites. In addition, the low cost of water and the price stability of large-scale procurement of raw materials make emulsion polymerization cost-effective from the raw material aspect. Currently, two major technologies are being expanded in the following directions: In catalytic green systems, the development of metal-free photocatalytic systems for ATRP is being focused on, and renewable resource-based monomers and emulsifiers are being explored for emulsion copolymerization. At the process control level, advanced sensing and data analysis technology are being used to achieve real-time monitoring of parameters to ensure the consistency of product quality. With respect to material applications, molecular design and process innovation are being utilized to prepare special structural composites to promote breakthroughs in electronics, medical and other fields. In summary, ATRP and emulsion polymerization technology, through mechanism innovation and process optimization, play key roles in the accurate synthesis and green production of polymers, and their cross-utilization will open up a broader space for the frontier research of functional materials.
Main Raw Materials. Methyl methacrylate (MMA), ethyl acrylate (EA), and 2-ethylhexyl acrylate (2-EHA) (analytically pure; Chengdu Cologne Chemical Co., Ltd., China) were used after vacuum distillation. Sodium dodecyl sulfate (SDS), pentamethyldivinyltriamine (PMDETA), deuterated dimethylsulfoxide (DMSO), ascorbic acid (AA), ethyl 2-bromoisobutyrate (EBIB), methanol (CH3OH), and potassium bromide (KBr) (analytically pure; Shanghai Aladdin Biochemical Science and Technology Co., Ltd., China) were also used.
Main Equipment and Instruments. A nuclear magnetic resonance spectrometer (WNMR-I, Tianjin Tester Instrument Co., Ltd., China), Fourier transform infrared spectrometer (FTIR920, Gongyi Yingyu Yarong Instrument Factory, China), recirculating water vacuum pump (SHZ-III, Zhengzhou Yuhua Instrument Manufacturing Co., Ltd., China), electrothermal constant-temperature drum and air drying oven (101-2AB, Tianjin Tianta Experimental Instrument Co., Ltd., China), precision timing electric stirrer (JJ-1, Changzhou Yingneng Experimental Instrument Factory, China), electronic analysis balance (AR2140, Shanghai Proanalytical General Instrument Co., Ltd., China), and constant-temperature bath (HH-WO-5L, Tianjin Top Instrument Co., Ltd., China) were used.
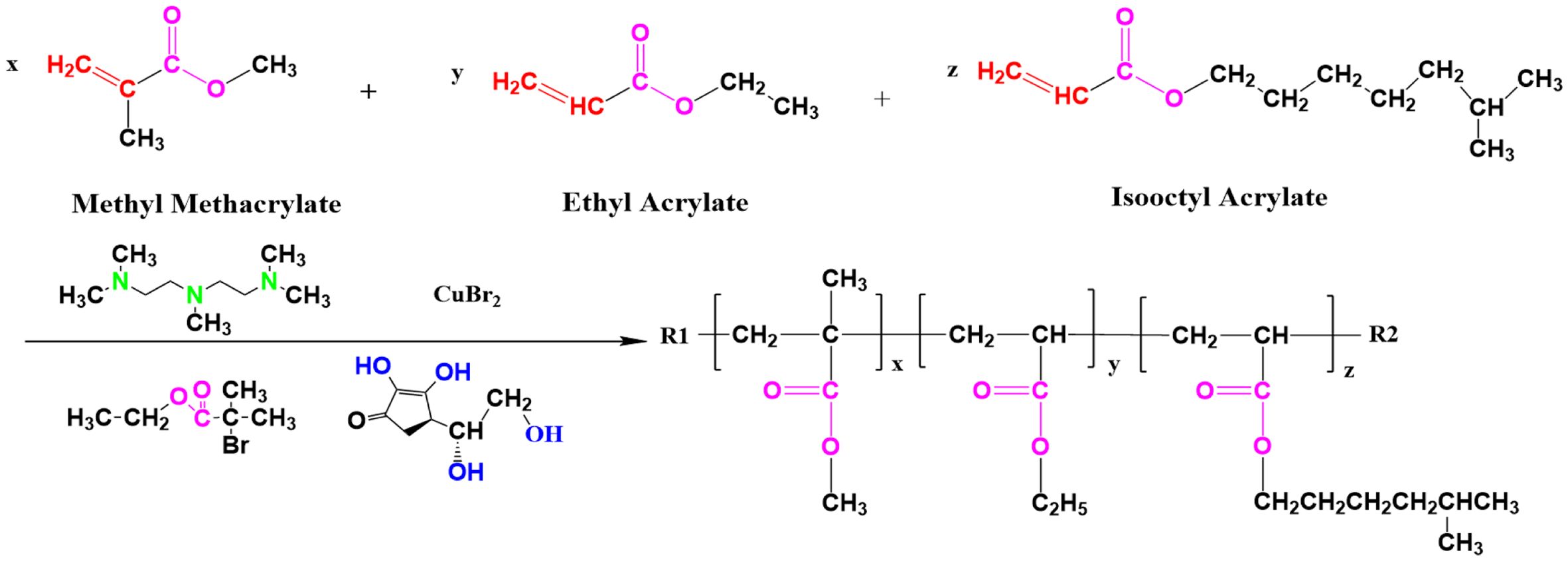
Synthesis of Acrylate Emulsions.To a 500 mL flask with a mechanical stirring device, a temperature detection device and a condensation reflux unit, 0.7 g of SDS and 140 mL of distilled water were added. The reaction temperature was adjusted to 60 ℃, and the stirring rate was 350 r·min-1 for 30 min to fully disperse the emulsifier in the aqueous phase. The temperature of the reaction system was increased to 80 ℃ while the stirring rate was adjusted to 300 r·min-1, followed by the addition of monomers, including MMA (24.5 g, 0.245 mol), EA (7.0 g, 0.07 mol), and 2-EHA (5.0 g, 0.0272 mol), as well as PMDETA and CuBr2 (0.075 g, 0.00034 mol), and the reaction was vented to nitrogen and maintained for 20 min. The mechanical stirring rate was adjusted to 240 r·min-1, the initiator EBIB was added, and the reaction time was controlled to 4 h after the emulsion was generated. Among the additives, n(EBIB):n(total monomer): n(CuBr2)=0.67:99.23:0.10. The results of the copolymerization reaction are shown in Figure 1.
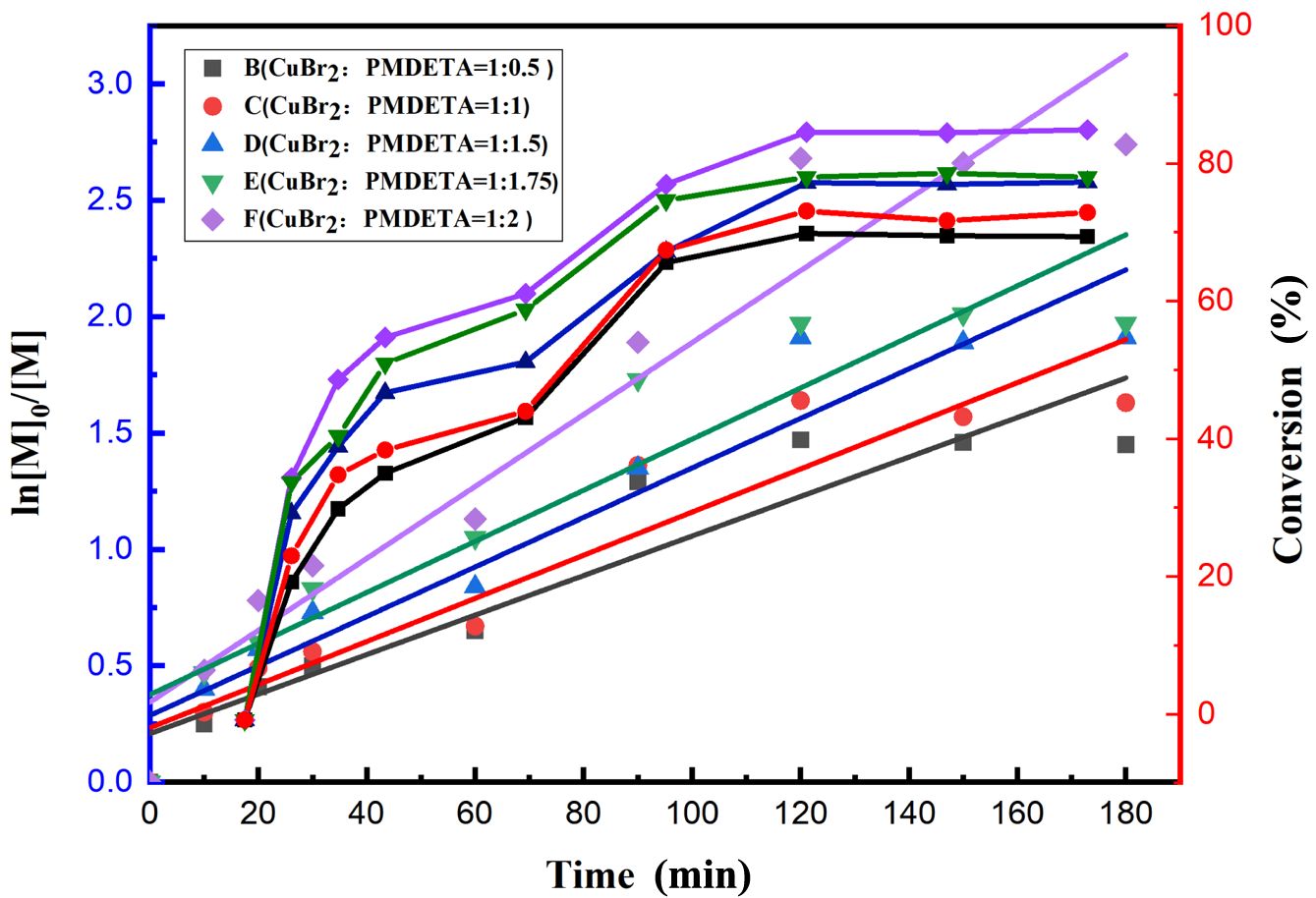
Analytical Methods. The conversion rate was determined by the gravimetric method. A 1 g emulsion sample was placed in a tin foil container, a small amount of a phenol ethanol solution was added to prevent reactions, and then, the sample was baked in a blast oven at 105 ℃ until a constant weight was reached. The monomer conversion can be calculated according to the following equation:
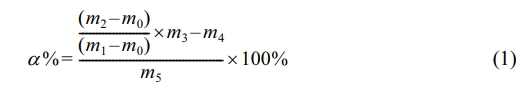
where m0 represents the mass of the tinfoil mold (g), m1 represents the mass of the sampled emulsion and tinfoil mold (g), m2 represents the mass of the dried sample and tinfoil mold (g), m3 represents the total mass of the emulsion (g), m4 represents the mass of the nonvolatile components of the emulsion (g), m5 represents the total mass of the monomers (g), and α% represents the conversion ratio.
Infrared Spectrum Analysis: The sample was dried at 27 ℃ in a blast drying box, the water was fully volatilized, and the remaining sample was ground into a powder. The sample was mixed with 5 mg of KBr, and the sample was analyzed by the fixed-angle specular reflection method. The wavenumber range was 400 cm-1 to 4000 cm-1.
Proton Nuclear Magnetic Resonance Spectrum Analysis: 1H NMR spectroscopy analysis reflects the nuclear magnetic resonance effect of 1H in a molecule. This method can be used to determine molecular structure. When a sample contains hydrogen, especially the isotope 1H, 1H NMR spectroscopy can be used to determine the structure of the molecule. The 1H isotope is also called protium. A simple hydrogen spectrum can be derived from a solution containing the sample. To avoid the interference of protons in the solvent, deuterated solvents are usually used. Deuterated dimethylsulfoxide (DMSO) was selected for this study.
|
Figure 1 ARGET–ATRP copolymerization reaction. |
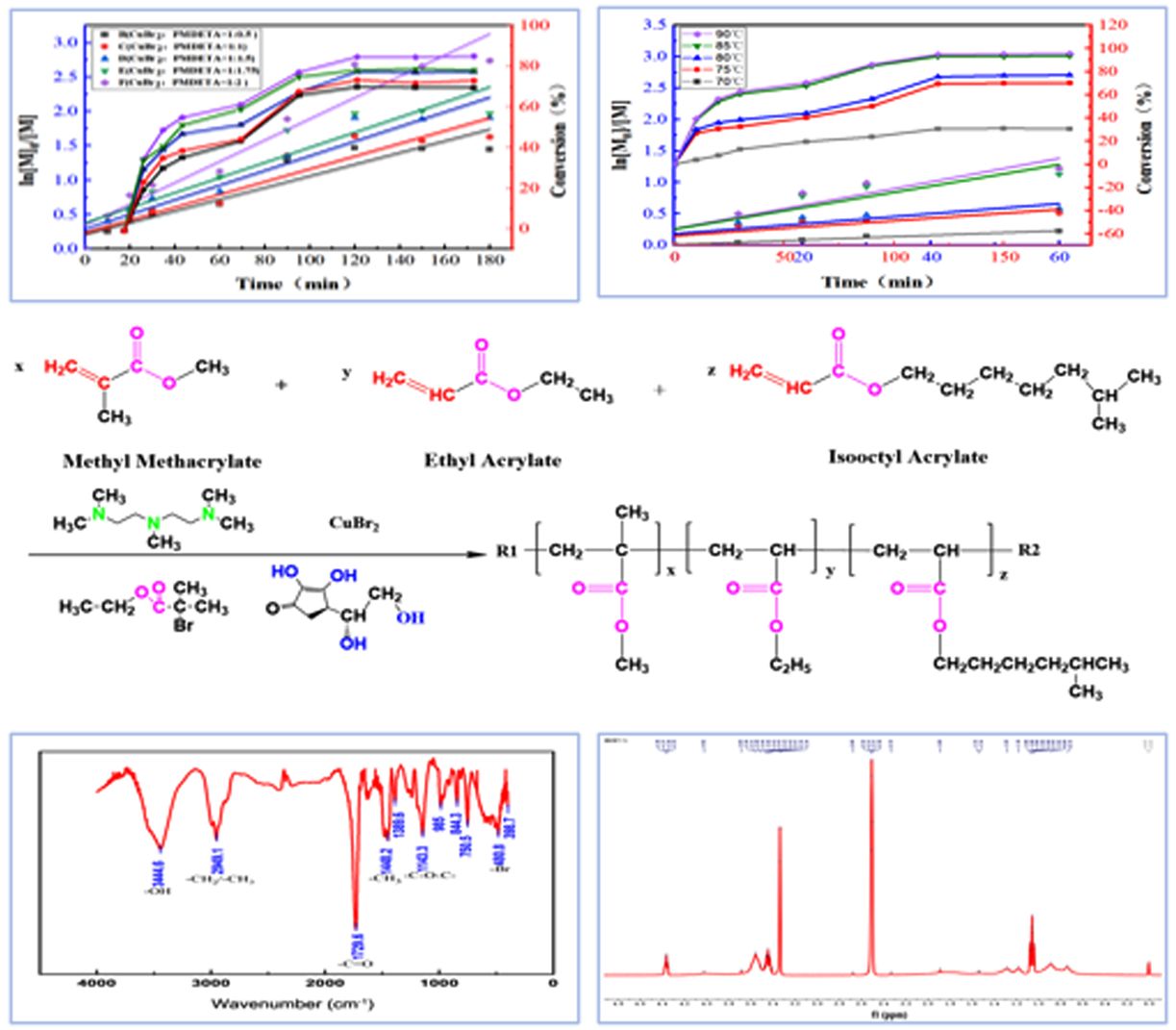
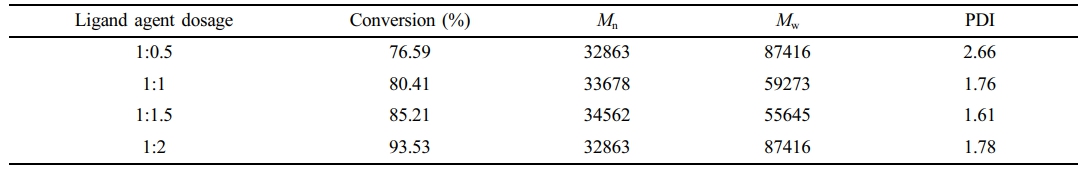
Influence of the Amount of Ligand Used in the Reaction System. During emulsion copolymerization involving metal catalysts, ligands can coordinate with metal ions, affecting the catalyst activity and selectivity, and play an important role in the reaction; thus, the amount of ligand to be added must be determined. To investigate the effect of the ligand PMDETA dosage on the copolymerization reaction, PMDETA-to-catalyst ratios of 0.5:1, 1:1, 1.5:1, 1.75:1, and 2:1 were selected for the study, and the effects of PMDETA content on the conversion rate are shown in Figure 2 (A point line graph, corresponding to the Y-axis on the right). This figure shows that the monomer conversion rate of each system continuously increased as the reaction proceeded. At the beginning of the reaction, the rate of increase was fast, but the rate gradually slowed and stabilized after 120 min, indicating that the reaction gradually approached equilibrium. Moreover, the conversion rate was significantly affected by the content of PMDETA and increased with increasing PMDETA content. When the ligand-to-catalyst ratio was 2:1, the conversion reached approximately 95%. This result occurred because the ligand formed complexes with the catalyst, which changed the electron cloud density and spatial structure of the catalyst. An appropriate increase in ligand input can effectively regulate the electron cloud distribution and spatial configuration of the catalyst active sites. This optimization can significantly increase the adsorption affinity of the catalyst for the reactants, promote the activation of reactant molecules, and increase the catalytic efficiency. In the late stage of the reaction, a small amount of monomers remained, which led to a slow increase in the conversion rate, which eventually leveled off. Since ligands directly affect the molecular weight of polymers, we further investigated the influence of different concentrations of ligands on the molecular weight. The test results are detailed in Table 1. The amount of ligand has a relatively small effect on the molecular weight and molecular weight distribution. When the ligand-to-catalyst ratio is 1:0.5 or 1:2, the molecular weight distribution is slightly greater. This increase occurs possibly because when a 0.5 molar ratio of ligand is added, the amount of ligand is lower, resulting in poorer control of the reaction. Moreover, at a high ratio of 1:2, the reaction conversion rate may be high, so the PDI is relatively large. However, at the proportion that can trigger the reaction to proceed, the amount of ligand has a relatively small effect on the molecular weight.
The effects of different ligand-to-catalyst ratios on the logarithm of the monomer concentration ln([M]0/[M]) are shown in Figure 2 (A scatter plot, corresponding to the Y-axis on the left). As shown in this figure, the corresponding copolymerization reaction rates sequentially increased in the order of ligand-to-catalyst ratios of 0.5:1, 1:1, 1.5:1, 1.75:1, and 2:1; with increasing ligand dosage, the reaction rate tended to increase. This phenomenon indicates that an increase in the amount of ligands helps increase the activity of the catalyst, thereby promoting the progress of the copolymerization reaction. Moreover, ln([M]0/[M]) was approximately linearly correlated with time, which was in accordance with the characteristics of first-stage reaction kinetics, indicating that the copolymerization reaction is first order. However, further observation revealed that the first-order kinetic curve had a slight curvature in the later stage of the reaction, indicating that the oxidation process of the catalyst might be progressive rather than a completely ideal first-order reaction.Among the ratios explored above, the apparent growth rate constant was the highest when the ligand-to-catalyst ratio was 2:1, implying that the polymerization rate is relatively fast in this case and that the reaction is superior. On the basis of these results, a catalyst:ligand ratio of 1:2 was selected for subsequent experiments.
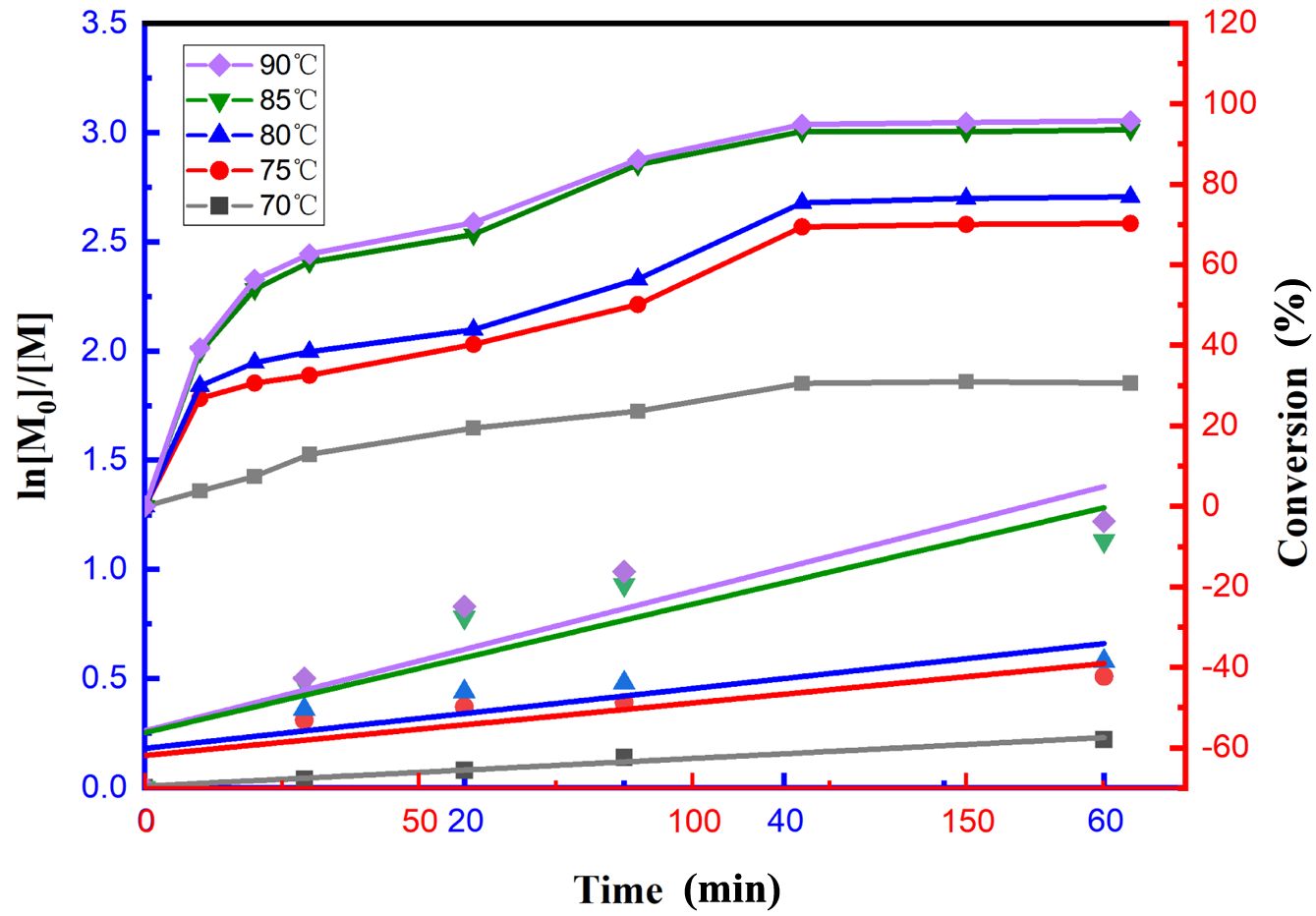
Effect of Reaction Temperature on the Reaction System.The copolymerization monomers used in the study were MMA, EA, and 2-EHA. The aforementioned catalytic system (CuBr2/PMDETA/EBIB) was used to copolymerize the monomer mixture. On this basis, five reaction temperatures, 70, 75, 80, 85, and 90 ℃, were used to study the effect of the reaction temperature on the reaction system. The correlation law between the temperature and conversion rate is shown in Figure 3 (point line graph, corresponding to the Y-axis on the right), according to which the conversion rate of the reaction increased with increasing reaction temperature, and the growth rate of the conversion rate in the early stage of the reaction was more considerable. When the temperature was 90 ℃, the conversion rate reached 95%, and at the same time, the higher the temperature was, the faster the increase in the conversion rate was, and the higher the conversion rate that could ultimately be achieved. These findings indicate that as the temperature increases, molecular thermal movement increases. Within the reaction system, the monomer molecules diffuse more rapidly, and their interactions with the active catalyst species become more frequent. This outcome facilitates the efficient propagation of polymer chains, resulting in an accelerated reaction rate and increased monomer conversion.
To investigate the kinetics of emulsion copolymerization, the conversion rates at different time points within 60 min were selected for study, and the effects of temperature on ln([M]0/[M]) are shown in Figure 3 (scatter plot, corresponding to the Y-axis on the left). This figure shows that ln([M]0/[M]) has an approximately linear correlation with time, which indicates that the emulsion copolymerization reaction conforms to first-order reaction kinetics. The corresponding polymerization rates (RPs) increased with increasing copolymerization temperature from 70 to 75, 80, 85, and 90 ℃, and the apparent growth rate constant was the highest at 90 ℃. With the gradual increase in the copolymerization temperature, the polymerization rate substantially increased. This phenomenon is consistent with the effect of temperature on the reaction rate, i.e., an increase in temperature leads to an increase in the effective collision frequency between molecules, which accelerates the copolymerization reaction.
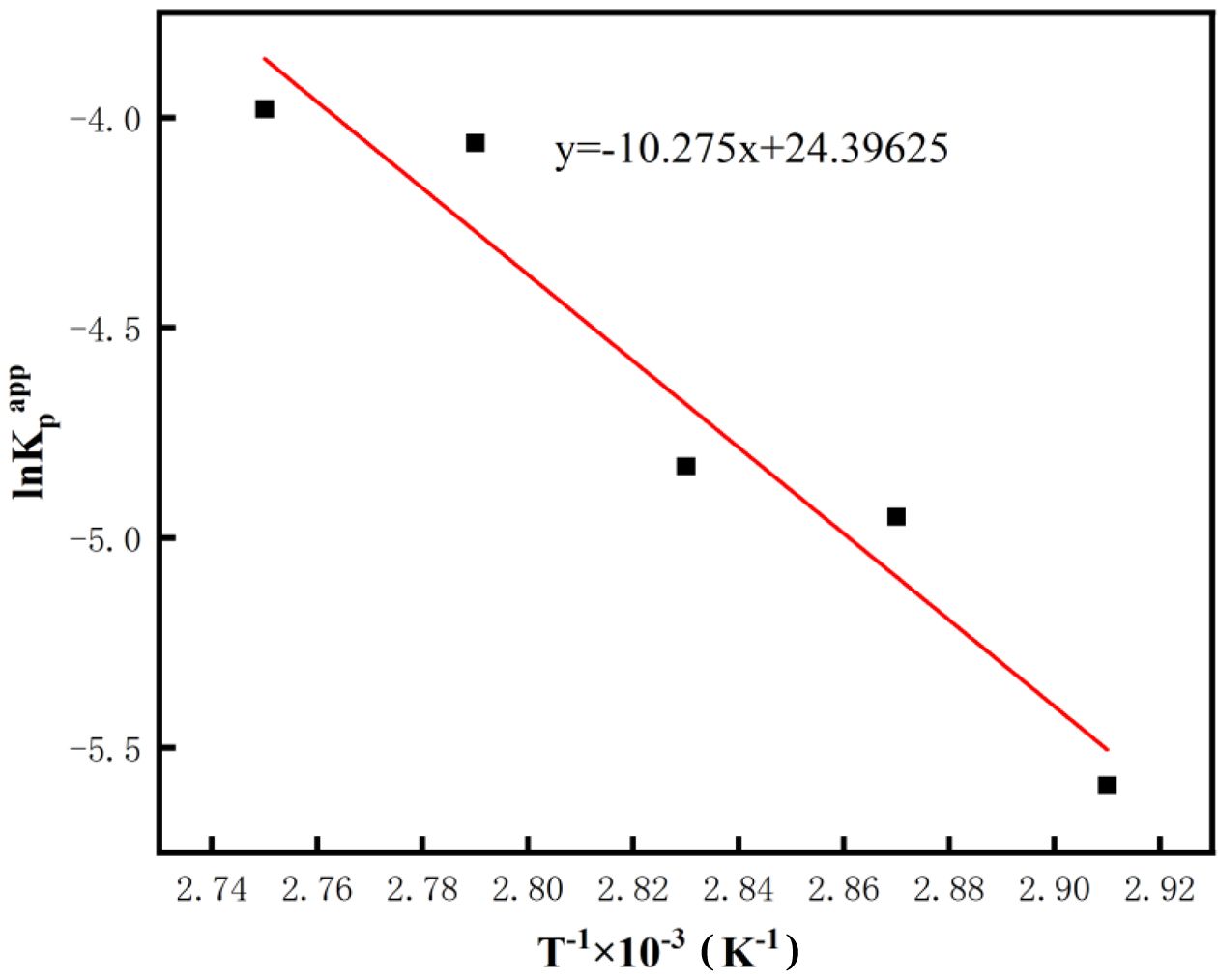
To further quantify the influence of temperature on the polymerization kinetics, the apparent polymerization rate constants (Kpapp) at different temperatures were analyzed based on the Arrhenius equation, as shown in Figure 4. Using the slope of the linear fit, the apparent activation energy Ea for this copolymerization reaction was calculated to be 80.43 kJ·mol-1. This provides a crucial theoretical foundation for a deeper understanding of the thermodynamic characteristics.
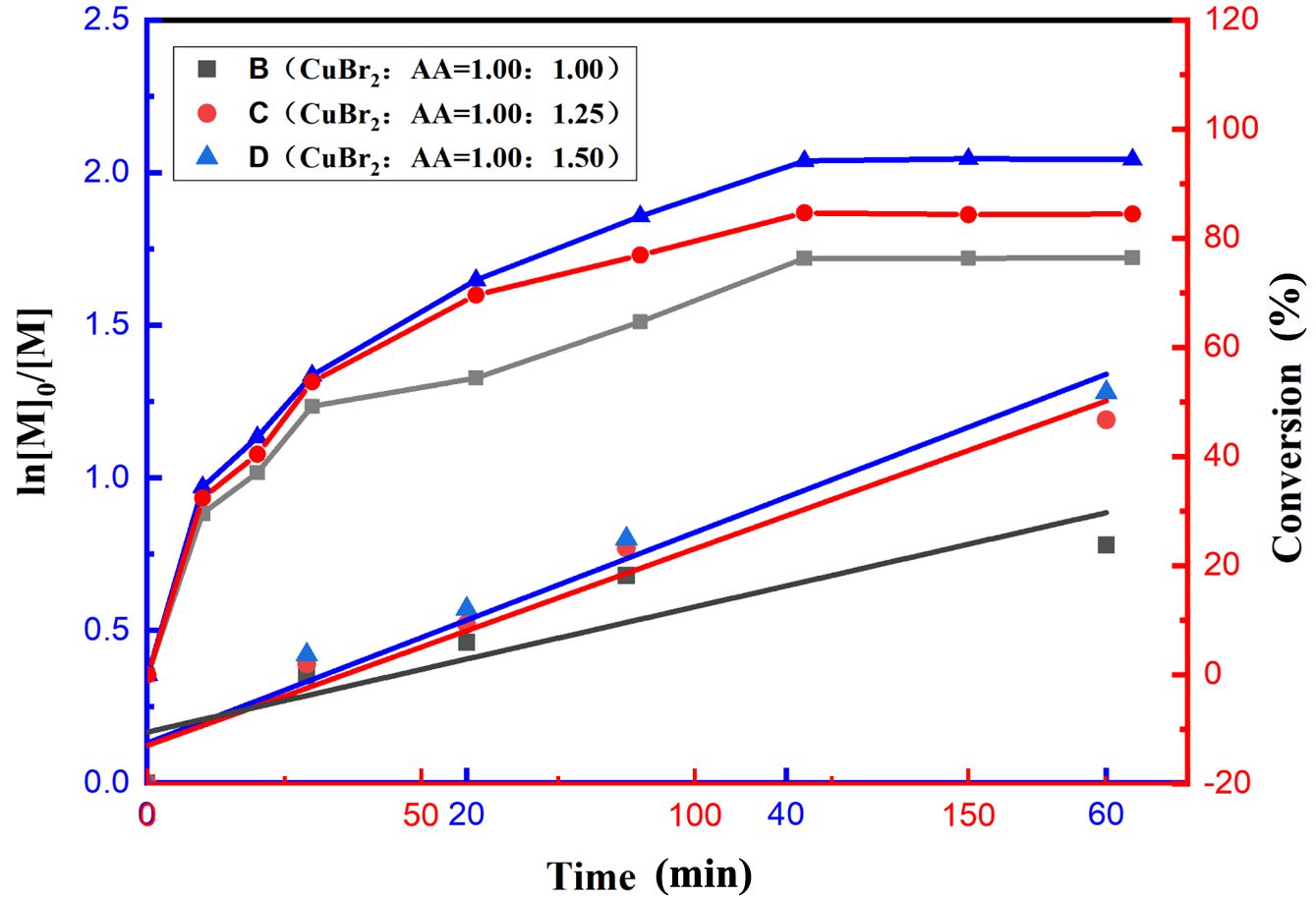
Effect of the Amount of Reducing Agent Used in the Reaction System. In the ARGET–ATRP process, the reducing agent can play an important role in converting high-valent transition metal salts into low-valent catalysts to sustain the catalytic cycle and maintain the copolymerization reaction. The aforementioned catalytic system was employed to study the effect of changes in the amount of reducing agent on the reaction system. At a constant copolymerization temperature of 85 ℃, the ratio of the reductant AA to the catalyst CuBr₂ was adjusted to investigate the effect of this ratio on the copolymerization reaction. The experimental data shown in Figure 5 indicate that variations in the reducing agent dosage considerably affect the logarithm of the monomer concentration ln([M]0/[M]) and polymerization conversion. The effect of the reductant dosage on monomer conversion is shown in Figure 5 (point line graph, corresponding to the Y-axis on the right), according to which the monomer conversion increased with increasing reductant dosage. These findings indicate that the variation in the reducing agent dosage in this system clearly affects the monomer conversion process. The effects of different reducing agent contents on the molecular weight and PDI of the polymers are shown in Table 2. As the AA-to-CuBr2 ratio increases, Mn increases slightly (from 31672 to 35236 g·mol-1), and the PDI decreases considerably (from 2.74 to 1.62).
The effect of the reducing agent dosage on ln([M]0/[M]) is shown in Figure 5 (scatter plot, corresponding to the Y-axis on the left). As shown in this figure, a good linear relationship exists for all the conditions. The concentration of the active centers was nearly constant throughout the copolymerization, which indicates that the polymerization process obeys first-order kinetics. The corresponding copolymerization reaction rate constants increased according to the following order of catalyst-to-reductant ratios: 1:1, 1:1.25, and 1:1.5. Notably, the rate of the polymerization reaction considerably increased when the amount of reducing agent per unit dosage increased. This phenomenon may be attributed to the increase in the concentration of AA enhancing its reducing activity toward Cu2+, which accelerates the production of low-valent copper ions, Cu(I), in the copolymerization system and thus increases the polymerization rate.
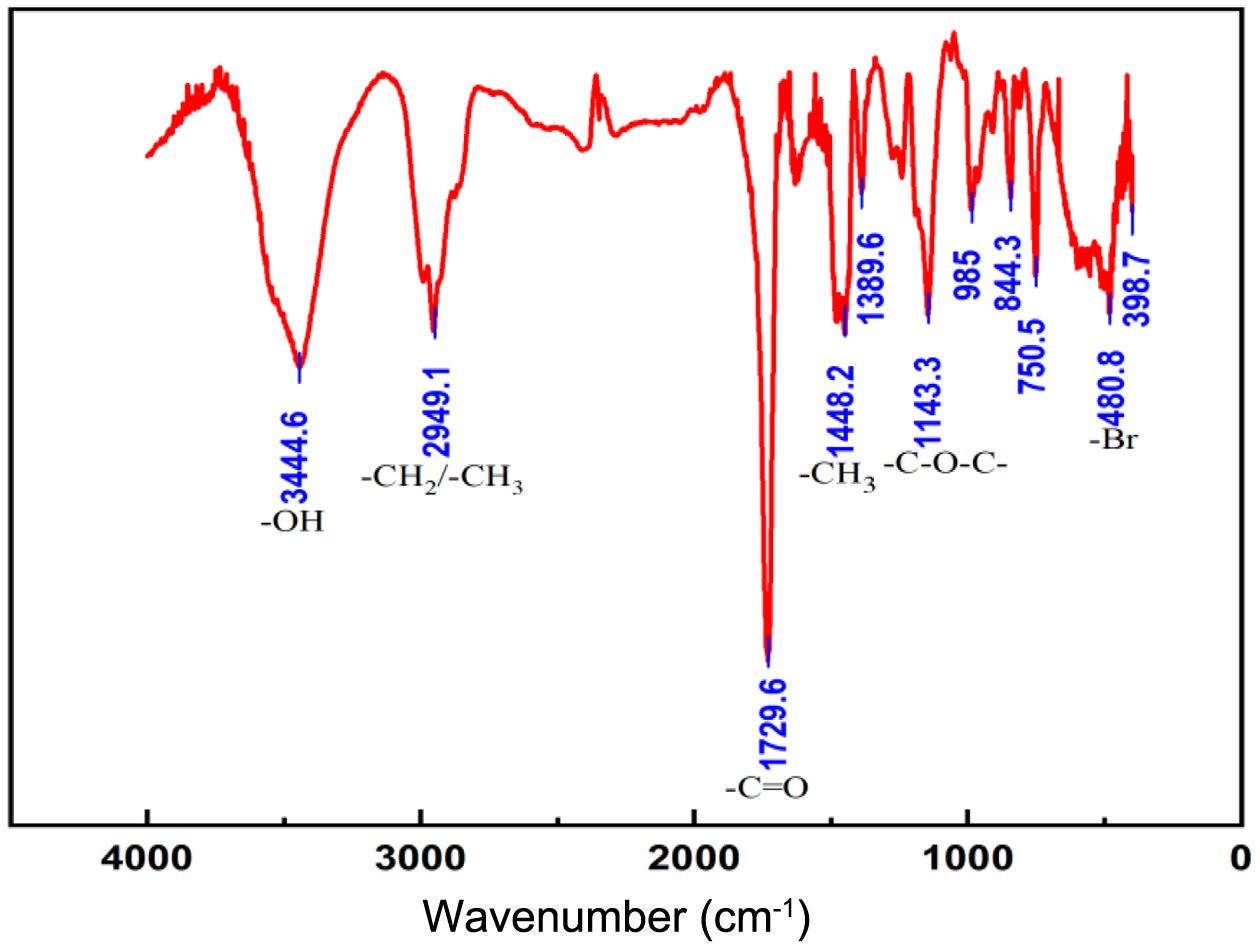
Characterization of the Polymers. Under specific experimental conditions, i.e., the catalyst-to-ligand molar ratio maintained at 1:2, the reaction temperature was kept constant at 90 ℃, and the catalyst-to-reductant molar ratio was set at 1:1.5; thus, a white powdery sample was successfully prepared. This sample was subsequently analyzed by infrared spectroscopy, and the results are presented in Figure 6. In the infrared spectra, the absorption peak at 3444.6 cm-1 was attributed to the stretching vibration of the hydroxyl group (-OH). The peak at 2949.1 cm-1 corresponded to the stretching vibration of the methyl group (-CH₃) and the methylene group (-CH2). The absorption peak at 1729.66 cm-1 originated from the stretching vibration of the carbonyl group (-C=O), which was highly consistent with the carbonyl character of the ester group (-COO-) in the structural formula. The methyl bending vibration peak, which occurred at 1448.2 cm-1, was assigned to the saturated alkyl group in the structural formula. The absorption peak at 1143.3 cm-1 corresponded to the stretching vibration of the ether bond (-C–O–C-). In addition, the characteristic peak at 480.8 cm-1 indicated the presence of bromine atoms; i.e., the product was capped with the initiator EBIB.
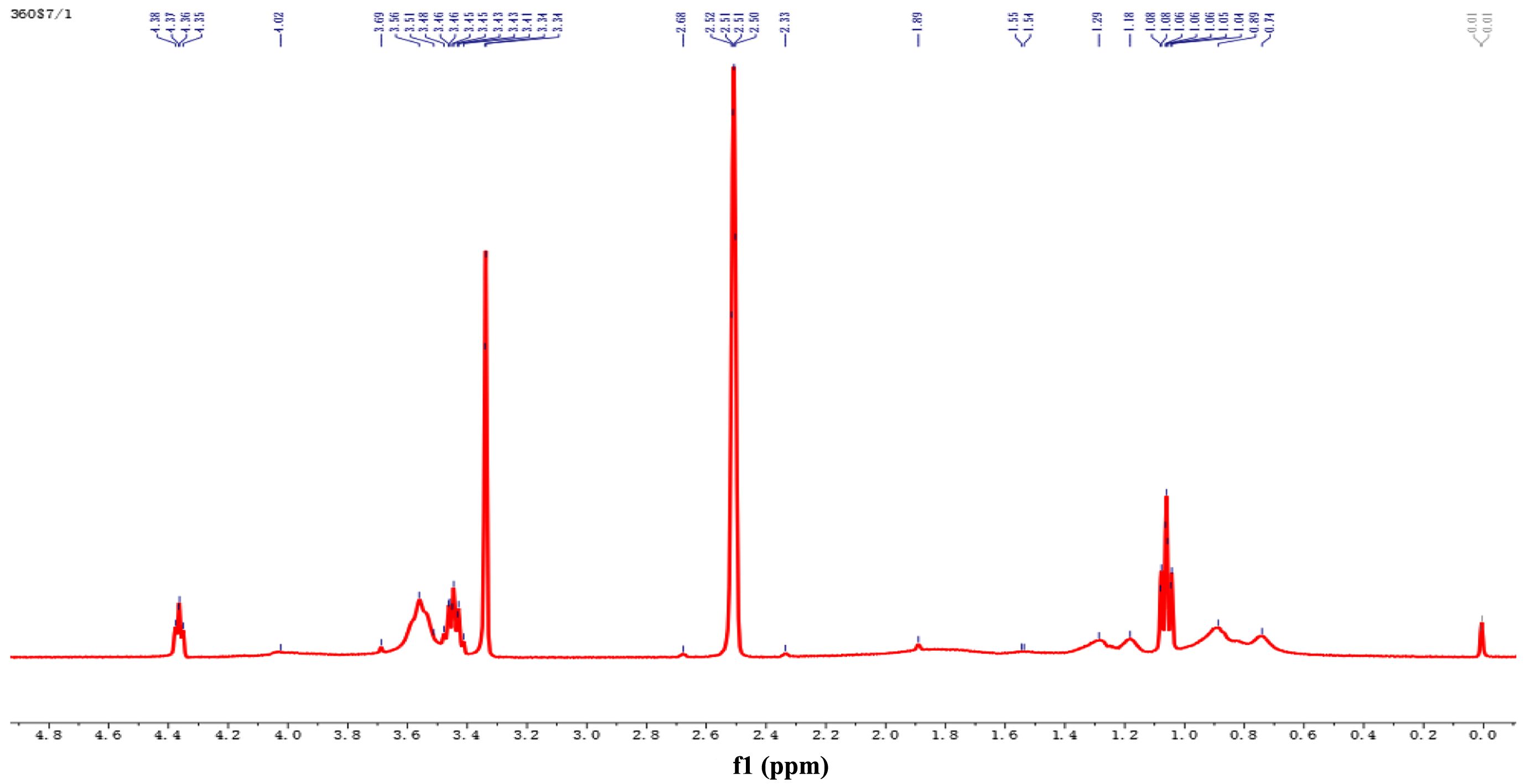
In the 1H NMR spectra, the chemical shifts reflect the chemical environment in which the hydrogen atoms are located. As shown in Figure 7, the peaks at δ » 0.8–1.2 ppm originated from the hydrogen atoms on the methyl group (-CH₃) in the alkyl group. The peaks in the range of δ » 1.2–1.4 ppm corresponded to the methylene hydrogens (-CH2-). The chemical shift peaks at δ » 3.4–3.6 ppm indicated hydrogen atoms attached to electronegative atoms, i.e., methylidene hydrogens in alcohols (-CH2OH) or ethers (-O–CH2-). The peak at δ » 4.6 ppm had a stronger chemical shift, i.e., a hydrogen atom in a more electronegative environment, so it corresponded to the hydrogen on the hydroxyl group (-OH) directly connected to the oxygen atom.
On the basis of the results of the 1H NMR experiments, the signal peak integral value ratio of the monomers MMA, EA and 2-EHA in the copolymer was 100:30.22:8.66, which was approximately the same as the experimental addition amount of 100:28.57:11.02. This finding indicates that all the monomers are effectively incorporated into the main chain of the copolymer and form a saturated chain structure. A comparison of the monomer conversion rate with the experimental results of nuclear magnetic resonance spectroscopy revealed that the integral value ratio was approximately consistent with the monomer feed ratio, further verifying the integrity of the copolymerization reaction and the uniformity of monomer incorporation.
|
Figure 2 Influence of the amount of PMEDTA used in the reaction system. |
|
Figure 3 Effect of reaction temperature on the reaction system. |
|
Figure 4 Functional correlation between the inverse temperature T-1 and the logarithmic value of the apparent rate constant lnKp app. |
|
Figure 5 Effect of the reducing agent amount used in the reaction system. |
|
Figure 6 Infrared spectra of the polymers. |
|
Figure 7 1 H NMR spectra of the polymers. |
In the present study, methyl methacrylate (MMA), ethyl acrylate (EA), and 2-ethylhexyl acrylate (2-EHA) were selected as the raw materials for the copolymerization reaction. Using activators regenerated by electron transfer–atom transfer radical polymerization (ARGET–ATRP), an acrylate emulsion was successfully prepared by changing the experimental conditions, such as the amount of ligand added, the temperature of the reaction system, and the amount of reducing agent, in a targeted manner. The synthesized copolymers were characterized and analyzed in detail using 1H NMR and FTIR spectroscopy in this study, and the results are as follows:
(1) As the amount of ligand increases, the monomer conversion rate tends to increase, and the rate constant of the copolymerization reaction accordingly increases, indicating that the amount of monomers promotes the copolymerization reaction. An increase in the reaction temperature can effectively increase the monomer conversion rate, and the two parameters are linearly related. After the kinetic calculations were performed, the apparent activation energy Ea of the emulsion copolymerization reaction was determined to be 80.43 kJ·mol-1, providing an important basis for an in-depth understanding of the thermodynamic properties of the copolymerization reaction. When the catalyst-to-reductant ratio was increased from 1:1 to 1:1.5, both the rate of the emulsion copolymerization reaction and the conversion of the monomers significantly increased, indicating that appropriate adjustment of the catalyst-to-reductant ratio helps optimize the performance of the copolymerization reaction.
(2) Analysis of the infrared spectra and proton nuclear magnetic resonance spectra clearly revealed the characteristic absorption peaks of the monomer, initiator and emulsifier, which fully proved that these components were involved in the copolymerization reaction and successfully formed the polymers, and these spectra provided strong spectroscopic evidence for the structure and composition of the products.
- 1. Kim, K.; Strong, J.; Sarkar, S. D.; Nguyen, D.; Dau, H.; Al-Aman, D. A. A.; Dadashi-Silab, S.; Harth, E.; Matyjaszewski, K. Polyethylene-poly(methyl acrylate) Block Copolymers from PACE-SARA ATRP: Utilizing Polyolefin Active Ester Exchange-Based Macroinitiators in Atom Transfer Radical Polymerization. Macromolecules 2025, 58, 1337-1348.
-

- 2. Yang, X. B.; Lei, T.; Tang, C.; Huang, X.; Wang, X. Y.; Huang, S.; Liu, X. Preparation and Characterization of Universal Cold-Resistant Reactive Chlorinated Acrylic Rubber and Study of Its Vulcanization Characteristics. Polym. Sci. Ser. B 2023, 65, 624-632.
-

- 3. Matyjaszewski, K. Atom Transfer Radical Polymerization (ATRP): Current Status and Future Perspectives. Macromolecules 2012, 45, 4015-4039.
-

- 4. Liu, X.; Li, Y.; Chu, Z. Y.; Fang, Y. C.; Zheng, H. L. Surface Modification of Bacterial Cellulose Aerogels by ARGET ATRP. J. Appl. Biomater. Funct. Mater. 2018, 16, 163-169.
-

- 5. Zhang, Z.; Wang, X. S.; Tam, K. C.; Sèbe, G. A Comparative Study on Grafting Polymers from Cellulose Nanocrystals via Surface-initiated Atom Transfer Radical Polymerization (ATRP) and Activator Re-generated by Electron Transfer ATRP. Carbohydr. Polym. 2019, 205, 322-329.
-

- 6. Dhar, A.; Koiry, B. P.; Haloi, D. Synthesis of Poly(methyl methacrylate) via ARGET ATRP and Study of the Effect of Solvents and Temperatures on Its Polymerization Kinetics. Int. J. Chem. Kinet. 2018, 50, 757-763.
-

- 7. Yang, W.; Hutchinson, R. A. The Influence of Adding Functionality to Dispersant and Particle Core Compositions in Non-aqueous Dispersion Polymerization. React. Funct. Polym. 2017, 114, 31-37.
-

- 8. Kwak, Y.; Magenau, A. J. D.; Matyjaszewski, K. ARGET ATRP of Methyl Acrylate with Inexpensive Ligands and ppm Concentrations of Catalyst. Macromolecules 2011, 44, 811-819.
-

- 9. Tanaka, K.; Matyjaszewski, K. Copolymerization of (Meth) Acrylates with Olefins Using Activators Regenerated by Electron Transfer for Atom Transfer Radical Polymerization (ARGET ATRP). Macromol. Symp. 2008, 261, 1-9.
-

- 10. Chan, N.; Cunningham, M. F.; Hutchinson, R. A. ARGET ATRP of Methacrylates and Acrylates with Stoichiometric Ratios of Ligand to Copper. Macromol. Chem. Phys. 2008, 209, 1949-1949.
-

- 11. Simakova, A.; Averick, S. E.; Konkolewicz, D.; Matyjaszewski, K. Aqueous Arget Atrp. Macromolecules 2012, 45, 6371-6379.
-

- 12. Król, P. Synthesis of PMMA-b-PU-b-PMMA Tri-block Copolymers Through ARGET ATRP in the Presence of Air. Express Polym. Lett. 2013, 7, 249-260.
-

- 13. Soly, S.; Mistry, B.; Murthy, C. N. Photo-mediated Metal-free Atom Transfer Radical Polymerization: Recent Advances in organocatalysts and Perfection Towards Polymer Synthesis. Polym. Int. 2021, 71, 159-1682021.
-

- 14. Zhou, J.; Chen, X.; Ma, J. Synthesis of Cationic Fluorinated Polyacrylate Copolymer by RAFT Emulsifier-free Emulsion Polymerization and Its Application as Waterborne Textile Finishing Agent. Dyes Pigm. 2016, 139, 102-109.
-

- 15. Singha, N. K.; Gibson, M. I.; Koiry, B. P.; Danial, M.; Klok, H.-A. Side-Chain Peptide-Synthetic Polymer Conjugates via Tandem “Ester-Amide/Thiol-Ene” Post-Polymerization Modification of Poly(pentafluorophenyl methacrylate) Obtained Using ATRP. Biomacromolecules 2011, 12, 2908-2913.
-

- 16. Beers, K. L.; Gaynor, S. G.; Matyjaszewski, K.; Sheiko, S. S.; Möller, M. The Synthesis of Densely Grafted Copolymers by Atom Transfer Radical Polymerization. Macromolecules 1998, 31, 9413-9415.
-

- 17. Zhao, C.; Harth, E. Polar Polyethylene Block Copolymer Synthesis via Organometallic-mediated Radical Polymerization Using Co (Salen) Complexes. Eur. Polym. J. 2024, 220, 113460.
-

- 18. Iqbal, A.; Fromann, L.; Saleem, A.; Ishaq, M. The Effect of Filler Concentration on the Electrical, Thermal, and Mechanical Properties of Carbon Particle and Carbon Fiber-reinforced Poly(styrene-co-acrylonitrile) Composites. Polym. Compos. 2007, 28, 186-197.
-

- 19. Yan, K.; Luo, Y. Significantly Suppressed Chain Transfer to Monomer Reactions in RAFT Emulsion Polymerization of Styrene. Ind. Eng. Chem. Res. 2019, 58, 20969-20975.
-

- 20. Zhu, Y. H.; Shen, Q.; Shen, F. Q.; Zhu, J. F.; Zhou, M.; Yao, C.; Yang, J. H. Synthesis of Organo-attapulgite/polyacrylate Nanocomposite Latex by Soap-free Emulsion Polymerization and Its Enhanced Film Properties. J. Coat. Technol. Res. 2025, 22, 1037-1047.
-

- 21. Lei, Q.; Peng, B. L.; Ma, K. K. Y.; Zhang, Z.; Wang, X. C.; Luo, J. H.; Tam, K. C. ARGET ATRP of Triblock Copolymers (PMMA-b-PEO-b-PMMA) and Their Microstructure in Aqueous Solution. ACS Omega 2018, 3, 15996-16004.
-

- 22. Kong, L. L.; Xu, D. D.; He, Z. X.; Wang, F. Q.; Gui, S. H.; Fan, J. L.; Pan, X. Y.; Dai, X. H.; Dong, X. Y.; Li, Y. F. Nanocellulose-Reinforced Polyurethane for Waterborne Wood Coating. Molecules 2019, 24, 3151-3151.
-

- 23. Cintora, A.; Köfer, F.; Yuan, C. Y.; Ober, C. K. Effect of Monomer Hydrophilicity on ARGET-ATRP Kinetics in Aqueous Mini-emulsion Polymerization. J. Polym. Sci. 2022, 60, 666-673.
-

- 24. Shao, M. L.; Yue, X. A.; Yue, T. Q.; He, J. Regulating Gelation Time and Kinetics Analysis Based on the ARGET ATRP Mechanism. J. Polym. Sci. 2020, 58, 519-527.
-

- 25. Krys, P.; Matyjaszewski, K. Kinetics of Atom Transfer Radical Polymerization. Eur. Polym. J. 2017, 30, 7901-7910.
-

- 26. Chen, M.; Zhou, H. W.; Zhou, L.; Zhang, F. Confined Polymerization: ARGET ATRP of MMA in the Nanopores of Modified SBA-15. Polymer 2017, 114, 180-188.
-

- 27. Yang, X. B.; Li, W.; Wu, Q.; Zhang, Q.; Li, J. Y.; Jiang, T. T.; Xiao, Q. Emulsion and Dynamics of Acrylate Prepared by ARGET-ATRP under a Compound Initiation System. Polym. Sci. Ser. B 2025, 66, 684-692.
-

- 28. Qiu, J.; Matyjaszewski, K. Polymerization of Substituted Styrenes by Atom Transfer Radical Polymerization. Macromolecules 1997, 30, 5643-5648.
-

- Polymer(Korea) 폴리머
- Frequency : Bimonthly(odd)
ISSN 2234-8077(Online)
Abbr. Polym. Korea - 2024 Impact Factor : 0.6
- Indexed in SCIE
 This Article
This Article
-
2026; 50(2): 244-252
Published online Mar 26, 2026
- 10.7317/pk.2026.50.2.244
- Received on Aug 26, 2025
- Revised on Nov 11, 2025
- Accepted on Nov 28, 2025
 Services
Services
- Full Text PDF
- Abstract
- ToC
- Acknowledgements
Introduction
Experimental
Results and Discussion
Conclusions
- References
Shared
 Correspondence to
Correspondence to
- Xingbing Yang*, ***, **** , Xinye Wang**
-
*College of Chemistry and Chemical Engineering, Sichuan University of Arts and Science
**Guangxi Key Laboratory of Electrochemical and Magnetochemical Function Materials,
College of Chemistry and Bioengineering, Guilin University of Technology
***Key Laboratory of Low-cost Rural Environmental Treatment Technology, Sichuan University of Arts and Science
****Special Polymer Materials for Automobile Key Laboratory of Sichuan Province, Sichuan University of Arts and Science - E-mail: yxb19830528@163.com, 3532264323@qq.com
- ORCID:
0000-0002-0254-6184, 0009-0005-4937-5406









 Copyright(c) The Polymer Society of Korea. All right reserved.
Copyright(c) The Polymer Society of Korea. All right reserved.