- Size-Controlled Polystyrene Microparticles via Thermodynamic Emulsion Polymerization
Department of Organic Materials and Textile Engineering, Jeonbuk National University, 567 Baekje-daero, Deokjin-gu, Jeonju-si, Jeonbuk 54896, Korea
*Biomedical Manufacturing Technology Center, Korea Institute of Industrial Technology, Yeongcheon 38822, Korea- 열역학적 에멀젼 중합에 의한 폴리스티렌 마이크로입자의 크기 제어
전북대학교 유기소재섬유공학과, *한국생산기술연구원 바이오메디칼생산기술센터
Reproduction, stored in a retrieval system, or transmitted in any form of any part of this publication is permitted only by written permission from the Polymer Society of Korea.
Controlled emulsion polymerization provides a well-defined and scalable route for synthesizing polystyrene (PS) submicron and nanoparticle dispersions with narrow size distributions. Owing to its ability to achieve high particle uniformity, tunable size control through thermodynamic and kinetic parameters, and compatibility with large-scale production, this technique has been extensively employed in both academic and industrial research. Nevertheless, conventional emulsion polymerization faces inherent limitations in producing micron-sized particles with a high degree of monodispersity, as particle growth is typically confined to the submicron regime. In this study, we systematically investigate the relationship between particle size distribution and key reaction parameters, including polymerization rate and initiator concentration, in emulsion polymerization. By invoking collision theory, we establish a quantitative correlation between particle size, initiator concentration, and reaction temperature, elucidating how these parameters govern nucleation, growth, and aggregation processes. Based on these insights, we develop a robust and systematic synthesis strategy that enables precise control over particle size across the 1–100 μm range by thermodynamically regulating nucleation, condensation, and particle–particle aggregation. Using this approach, we successfully demonstrate the targeted synthesis of monodisperse PS particles with diameters approaching ~100 μm.
에멀젼 중합은 좁은 입도 분포를 갖는 폴리스티렌(PS) 서브마이크론 및 나노입자 분산체를 대량 합성하는 데에 활용된다. 높은 입자 균일성을 확보할 수 있고, 열역학적·동역학적 변수 조절을 통해 입자 크기를 조정할 수 있으며, 대량 생산 공정과의 호환성이 뛰어나다는 장점으로 인해 에멀젼 중합은 학계와 산업계 모두 폭넓게 활용되어 왔다. 그러나 기존의 에멀젼 중합은 입자 성장이 일반적으로 서브마이크론 영역에 제한되기 때문에, 높은 단분산성을 유지한 마이크론 크기 입자를 제조하는 데에는 본질적인 한계를 지닌다. 본 연구에서는 에멀젼 중합 과정에서 입자 크기 분포와 주요 반응 변수(중합 속도 및 개시제 농도 등) 간의 관계를 체계적으로 살펴보았다. 또한 충돌 이론(collision theory)을 적용하여 입자 크기, 개시제 농도, 반응 온도 사이의 정량적 상관관계를 도출하고, 이러한 변수들이 입자 합성의 핵생성(nucleation), 성장(growth), 응집(aggregation) 과정에 어떻게 작용하는지를 규명하였다. 이를 바탕으로 핵생성, 응축(condensation), 입자 간 응집을 열역학적으로 제어함으로써 1–100 μm 범위에서 입자 크기를 정밀하게 조절할 수 있게 되었다. 또한, 이 방법을 활용하여 직경이 약 100 μm에 근접하는 단분산 폴리스티렌 입자의 합성에 성공하였다.
We systematically analyzed how polymerization rate, initiator concentration, and temperature affect particle size distribution in emulsion polymerization. Using collision theory, we established quantitative relationships among these parameters and particle size, clarifying their roles in nucleation, growth, and aggregation. Based on this understanding, we developed a thermodynamically controlled synthesis strategy that enables precise size control from 1 to 100 ¥ìm and demonstrated the production of monodisperse polystyrene particles up to ~100 ¥ìm in diameter.
Keywords: emulsion polymerization, particle size distribution, initiator, thermodynamics, aggregation.
This paper was supported by research funds for newly appointed professors of Jeonbuk National University in 2024.
The authors declare that there is no conflict of interest.
Information is available regarding IR spectrums of all synthesized PS particles, topographic chart of size distribution with all the conditions of temperature and initiator concentration, statistical data of the size of synthesized PS particles, synthesized PS particles solid concentration per time, and size distribution diagrams of all centration conditions. The materials are available via the Internet at http://journal. polymer-korea.or.kr.
PK_2026_050_02_295_Supporting_Information.pdf (355 kb)
Supplementary Information
The size-controlled synthesis of micro/nano-polystyrene (PS) particles has been extensively explored due to its diverse applications in various industrial sectors, including the bio-industry,1 composite materials,2 and energy technologies.3 Micro/nano-PS particles serve various functions, such as carriers for drug delivery systems,2 and scattering media in cosmetic formulations.4 Consequently, controlling the size of PS particles is of paramount importance as it affects their surface area to volume ratio and performance in numerous applications. Emulsion polymerization5 is a well-established technique for synthesizing sub-micron PS particles, while precipitation polymerization is commonly used for larger PS particles, typically around ~10 μm.6 In addition, suspension polymerization is used for particles around ~sub mm in size.7 However, it is difficult to achieve an optimal synthesis regime in the intermediate size range of about ~50 μm. This is because it is extremely challenging to control particle growth in this size range.
Particle growth by polymerization involves three fundamental steps: nucleation,8 condensation,9 and agglomeration.10 Precisely controlling these processes one at a time to achieve the specific particle size is extremely difficult because these three steps occur simultaneously growing particles exponentially. Previous studies have made significant progress in optimizing particle size using emulsion polymerization, mainly controlling the nucleation and condensation steps for size uniformity by inhibiting agglomeration with surfactant; this method is preferable to synthesize less than 1 μm particles.11 Particles with ~100 micron in size are difficult to grow by condensation alone. Agglomeration occurs by collisions between particles, which leads to exponential particle growth, however, it is very difficult to control the collision frequency to reach out ~100-micron particle size with uniformity. The significance of ~100-micron particles lies in one of its potential applications such as in chromatography resins. Chromatography is a crucial technique in various fields, particularly in drug purification and characterization.12 The use of micro-sized particles, specifically designed and controlled in shape, can enhance the efficiency and performance of chromatography resins. The ~100-micron size range is particularly relevant because it strikes a balance between being large enough to offer advantages in terms of flow dynamics and separation efficiency,13 yet still small enough to retain the benefits of increased surface area.14 This makes these particles ideal candidates for creating advanced chromatography materials that can contribute to more precise and effective separation processes in diverse scientific and industrial applications.
In our approach to obtain ~100 µm size particles, a systematic synthesis method has been developed for tailoring desired particle sizes between 1-100 µm by thermodynamically manipulating nucleation, condensation, and agglomeration. It begins with the creation of seed particles (nucleation), initiated by a thermal initiator, serving as the foundation for subsequent growth by agglomeration. In addition, significant control over the final particle size is achieved by modulating the collision frequency by manipulating key thermodynamic parameters such as temperature and monomer concentration. This strategy allows the growth process to be fine-tuned, ensuring precise achievement of desired sizes with high yields. To be specific, PS particles in the 50-100 µm size range were synthesized. The adaptability introduced by modulating these conditions enhances the reproducibility and broadens the applications across industries, from biotechnology to materials science. This approach not only achieves monodisperse particle synthesis, but also prioritizes the thermodynamic mechanism over agglomeration, enabling precise size control within the desired range of 1 to 100 µm. This dynamic growth process not only allows precise size distribution control but also opens up new possibilities for scalability, providing a promising avenue for applications where a broad range of particle sizes is crucial.
Materials. Styrene (>99%, Sigma-Aldrich, lot #STBK2797, Germany), P123 (Sigma-Aldrich, Germany), potassium persulfate (>99%, Sigma-Aldrich, Germany), ethanol (95%, Daejung Chemicals, South Korea), distilled water (Daejung, South Korea) and Nitrogen gas (99%, South Korea) was used for the experiments.
Preparation of PS Micro particles. In a 250 mL flask, 0.1 g of P123 (PEG-PPG-PEG block copolymer) was dissolved in 90 mL of distilled water under gentle heating (65 ℃). Then 10 mL of styrene was added to this preheated solution and mixture was stirred at 1500 rpm for 30 minutes. Subsequently, 0.1 g of potassium persulfate dissolved in 10 mL of distilled water was added, initiating the polymerization under reflux at 65 ℃ for 5 hours. To further drive the reaction, an additional 0.5 g of potassium persulfate in 10 mL of water was added, followed by continued reflux for 1 hour. The entire process was conducted under an inert nitrogen atmosphere.
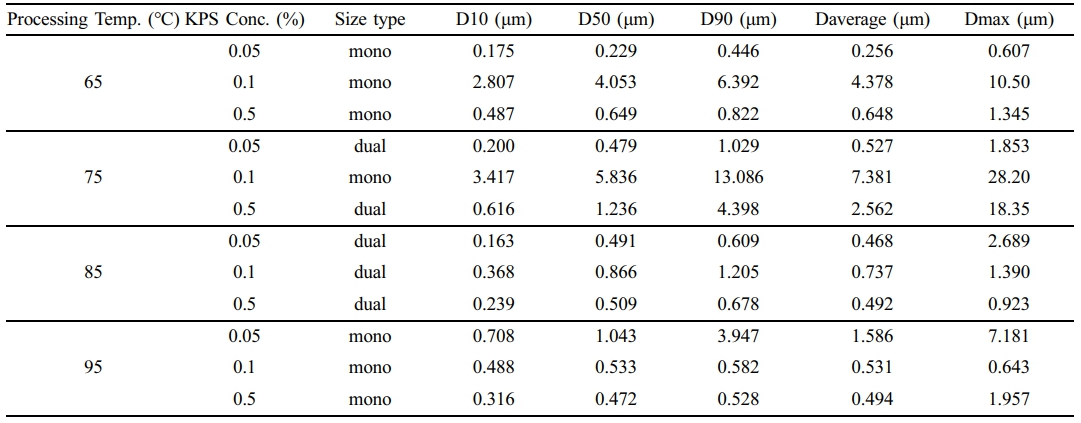
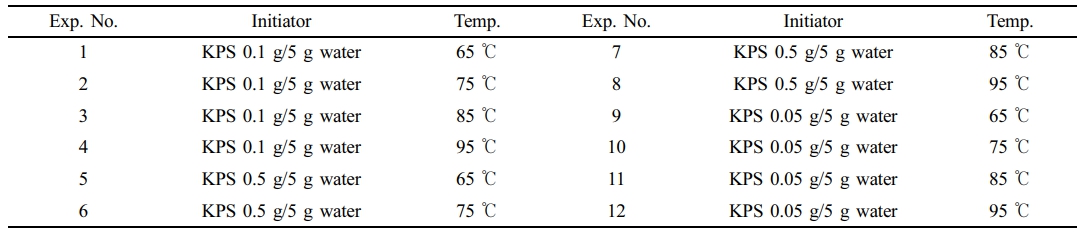
The versatility of our synthesis method was investigated by repeating the procedure 11 times with varying temperatures and potassium persulfate concentrations. All experiments employed a consistent protocol using 0.1 g P123, 10 mL styrene, 90 mL water, and stirring at 1500 rpm for 5 hours. Details of the temperature and concentration variations are provided in Table 1.
Extraction of PS Microparticles. PS microparticles were extracted via temperature-controlled precipitation, followed by addition of 300 mL ethanol and vigorous stirring for 30 minutes. Ultrasonic dispersion for 1 hour facilitated further extraction, and centrifugation at 4000 rpm for 15 minutes isolated the solid phase, which was then redispersed in ethanol using ultrasonic agitation. Filtration through a 200-mm sieve eliminated chunky impurities. Finally, the solid phase was washed with ethanol and stored in the same solvent at room temperature.
Characterization. The Fourier Transform Infrared (FTIR) spectrophotometer (PerkinElmer Spectrum One) was employed to verify successful polymerization and identify PS micro particles. Scanning electron microscopic analysis was conducted employing the Genesis-2020 SEM to analyze the morphology of PS micro particles.
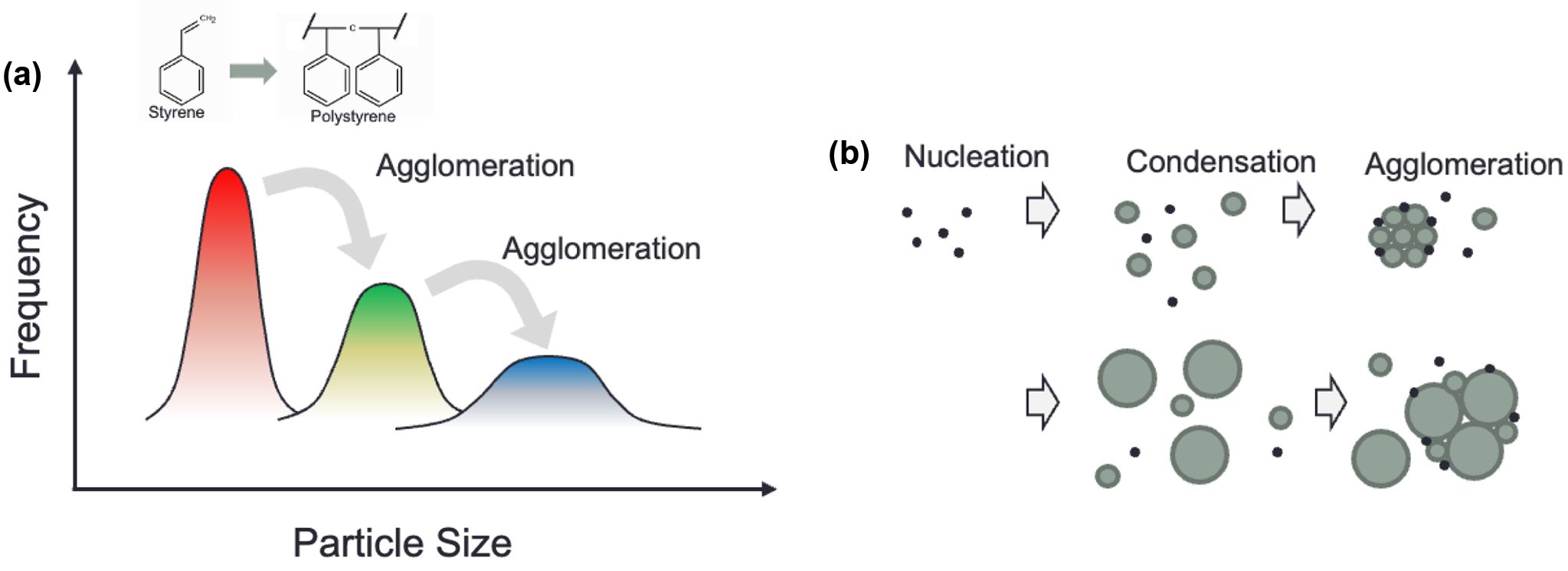
In this work, the particles undergo a series of stages involving nucleation, condensation, and agglomeration, as shown in Figure 1(a)-(b). This aggregation process repeats numerous times during the reaction, leading to an exponential increase in particle size. This complexity makes it a challenge to precisely control the exact size of the final particle. Particle growth mainly involves two mechanisms: condensation and agglomeration. After nucleation, particles gradually grow by polymerizing monomers, undergoing moderate growth. Subsequently, they collide to other particles, leading to a sudden increase in volume, shown in Figure 1(a). The particles subsequently agglomerate by forming larger particles. P123 was employed as a nonionic surfactant for emulsification during polymerization. Owing to its nonionic nature, P123 reduces electrostatic repulsion between particles and thereby promotes particle aggregation, in contrast to ionic surfactants, which induce strong interparticle repulsion and inhibit aggregation. A fixed amount of P123 was initially evaluated, and the amount of surfactant was decided by systematically increasing the surfactant concentration until stable particulate dispersions were obtained, rather than macroscopic polymer chunks, during polymerization.
An in-depth experimental examination has been performed to optimization of the particle size with the variation of temperature, initiator percentage, and monomer concentration. Specifically, increasing the temperature, a key factor for thermal initiator, accelerates the decomposition and activation of the initiator. This leads to a higher concentration of free radicals in the environment, which in turn enhances the rate of nucleation.
Scanning electron microscopic images of PS microparticles synthesized under 12 various conditions of temperature and potassium persulfate (KPS) concentration shown in Figure 2. Statistical analysis of the SEM image of PS-particle diameters revealed a complex interplay between these parameters. Particle size increased with temperature up to 85 ℃, followed by a decrease at higher temperatures. Similarly, KPS concentration exhibited a non-linear effect. Particles remained around 1 μm at 0.05 g (0.5 wt%) KPS, increased to 10 μm at 0.1 g (1%), and then decreased back to 1 μm at 5 g (5%). Statistical analysis of the size of the synthesized PS particles under different condition. As shows in Table 2, demonstrating the effectiveness of the optimization process with the temperature and initiator concertation. FTIR spectra of all synthesized PS particles shown in Figure S1, which shows that the formation of the PS particles depending on the reaction temperature and the concentration of the initiator (KPS). This suggests that the also reaction conditions can affect the chemical structure of the PS particles leads to formation of different size of the particle. For a deeper understanding of this mechanism, the Smith-Ewart Theory elucidates how particle size in emulsion polymerization is influenced by the interplay between nucleation and condensation events, which relates back to the fundamental concepts of nucleation, condensation, and agglomeration. Increasing the concentration of initiators promotes nucleation (providing more starting points for particles), resulting in smaller particles. However, a higher polymerization rate can have a dual effect. Faster growth (condensation) of existing particles can lead to smaller final sizes. Conversely, a high polymerization rate can also initiate numerous short chains that compete for monomers, potentially resulting in larger particles through agglomeration (collisions and merging). Although the theory does not explicitly address agglomeration, additional factors such as initiator amount and the interaction between propagation and termination rates (influenced by temperature) further refine the understanding of how these processes affect particle size and distribution. Therefore, considering all these factors, applying the Smith-Ewart Theory, we can understand and predict how changes in initiator concentration and temperature affect particle formation and size in emulsion polymerization.
The growth of polymer particles in dispersion systems can be effectively described by collision theory, which focuses on the frequency and consequences of binary collisions between particles. In particular, when two identical polymer particles collide, the collision frequency (Z) is given by:

where, n is the number density of polymer particles, r is the radius of each polymer particle, kB is the Boltzmann constant, T is absolute temperature and m is mass of a polymer particle. The rate of particle size growth is strongly dependent on the collision frequency. An increase in temperature enhances thermal mobility of particles, thereby increasing the collision frequency and promoting larger particle growth. In contrast, the concentration of initiatordetermines the initial number of polymer nuclei formed. A higher initiator concentration results in the formation of a greater number of smaller initial particles. This increases the number density (n) (Eq. 1), raises the collision frequency. Thus, the initiator concentration can be optimized to maximize the collision frequency, balancing the trade-off between particle number and size. Furthermore, temperature not only affects the kinetics of particle collisions but also plays a critical role in initiator activation. For example, potassium persulfate (KPS) exhibits significant activation at approximately 65 ℃. Therefore, temperature serves a dual purpose: 1) Enhancing collision frequency thermodynamically and 2) Triggering initiator activation to increase the number of particles. By tuning both the initiator concentration and temperature, the system can be optimized for maximumparticle growth. This illustrates how particle size evolution during polymerization is jointly governed by thermal and chemical parameters.
Both condensation and aggregation processes also depend upon temperature in the polymerization process. For condensation polymerization, lower temperatures (closer to 60 ℃) favor larger particles due to slower reaction kinetics, while higher temperatures (closer to 90 ℃) favor smaller particles due to higher reaction kinetics. But in the agglomeration model, higher temperatures (closer to 90 ℃) promote faster aggregation and larger particle sizes, whereas lower temperatures (closer to 60 ℃) lead to smaller particles. So, explaining the above equation for each model, the synthesized PS-particle size can be control by adjusting the initiator concentration and temperature accordingly. The Figure S2 shows that the topology chart of the size distribution with the variation of temperature and initiator concentration. Figures S2(a) and S2(b) illustrate the size of the first and ninth decile particles respectively. Both the figures show that as the processing temperature increases from 65 ℃ to 95 ℃, yield percentages also increase, with more pronounced changes observed at higher KPS concentrations (0.5% KPS). Specifically, at lower temperatures (65 ℃), yield is smaller across all KPS concentrations, whereas at higher temperatures (95 ℃), yield is significantly higher, especially at 0.5% KPS. This trend is consistent in both the first and ninth decile particle sizes, emphasizing the impact of temperature and initiator concentration on yield percentage.
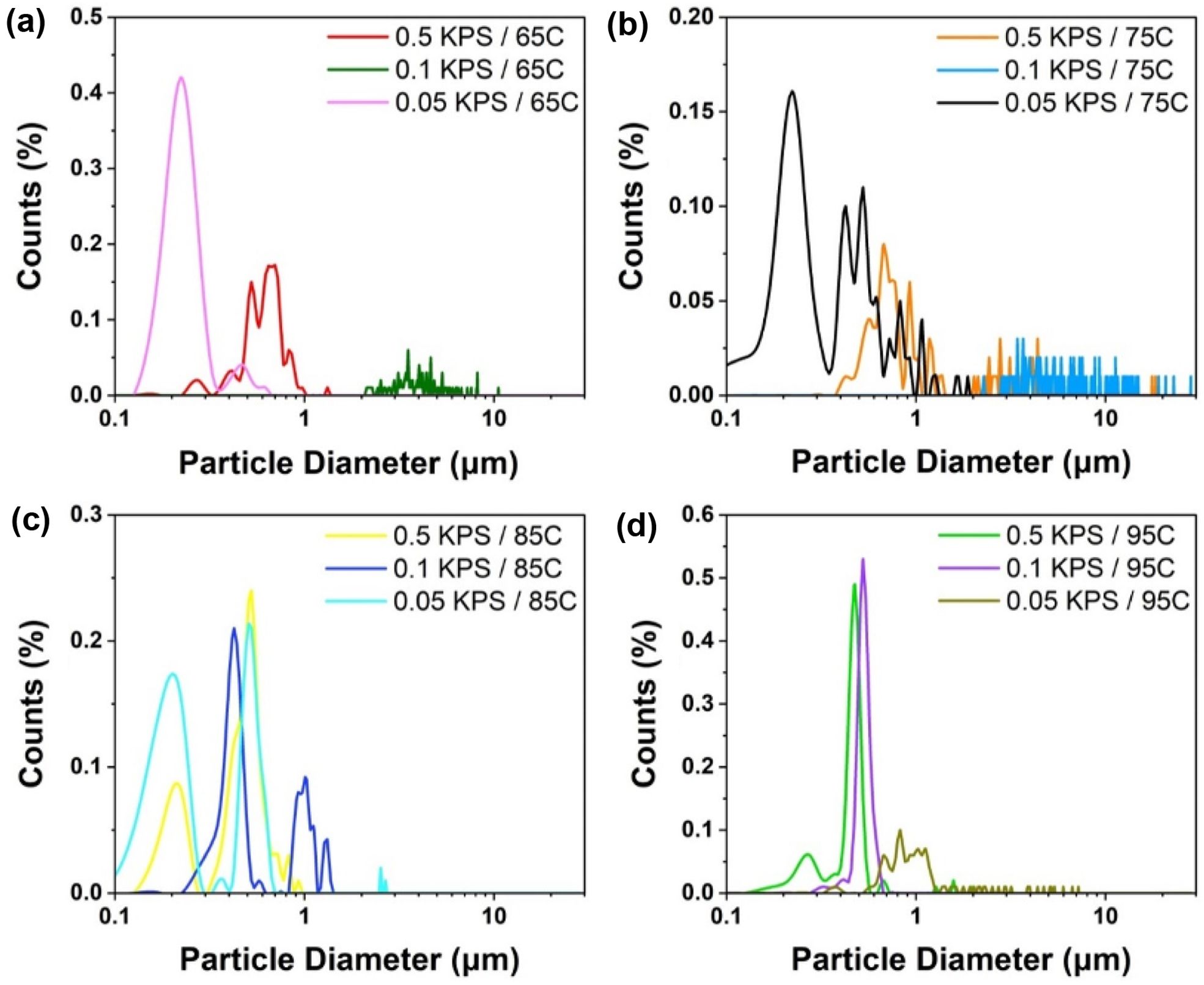
To better understand the effect of the initiator system with the variation of temperature on the particle coagulation and particle size distribution, the KPS system was first considered. The KPS initiator system is one of the most widely used initiators in conventional emulsion polymerization in pure water solvent.15 The theory of Smith–Ewart (micellar theory) predicted a proportionality between particle number and KPS initiator concentration. These theories confirmed the relationship between the particle size and initiator concentration. The particle size distribution of synthesized PS-particles under different initiator concentrations (KPS) with varying thermal condition is shown in Figure 3. At 65 ℃, Figure 3(a) shows that particle formation with 0.05% KPS have the smallest size and narrowest distribution, while the largest range of particle sizes, exceeding 10 µm, formed by 0.1% KPS. The particle size distribution shifted towards larger particles for all KPS concentrations as the temperature increase to 75 ℃, as shown in Figure 3(b). It is interesting to note that 0.5% and 0.1% KPS shows higher size distribution than those of 0.05% KPS. When the temperature reaches at 85 ℃, as shown in Figure 3(c), the distributions become more concentrated around smaller particle sizes. Particle size distributions for all KPS concentrations reduced even further at 95 ℃ and peak count percentages rise, indicating more uniform and smaller particles. The smallest and most uniform particles are produced by 0.1% KPS. To further explanation for the optimization of initiator concentration and temperature for the synthesis of PS-particles, solid concentration compares to Sphere particles yield in different condition, which shows in Figure S3. Figure S3(a) suggests that the PS-particles produced have a range of sizes, and the distribution of these sizes can be affected by the reaction conditions, such as polymerization initiator concentration (KPS) and temperature. The particles synthesized at lower polymerization rates (0.1 KPS) tend to be smaller than the particles synthesized at higher polymerization rates (0.5 KPS). This is likely because there are fewer initiator molecules available at lower polymerization rates, which leads to slower growth of the PS particles. The synthesized PS-particles solid concentration increases with the time as shown in Figure S4. As the reaction proceeds, more monomers convert into polymer chains, increasing the mass of solid PS-particles within the system. Initial rate of polymerization is high due to the availability of a large number of monomers. As the reaction progresses, the decrease in monomers concentration slow down the rate, but the overall mass of the polymer continues to increase. Figure S3(b) shows that, at a KPS concentration of 0.1 and a temperature of 75 ℃, the graph shows a broad peak around 10 µm particle diameter, indicating a lower percentage of particles in that higher size range. Figure S5 shows that, at higher concentrations (0.5% KPS), the particle size distribution is narrower with a peak around 1 µm. As the concentration of KPS decreases (0.1% and 0.05% KPS), the particle size distribution broadens, with a larger proportion of particles exceeding 3 µm with the increasing temperature.
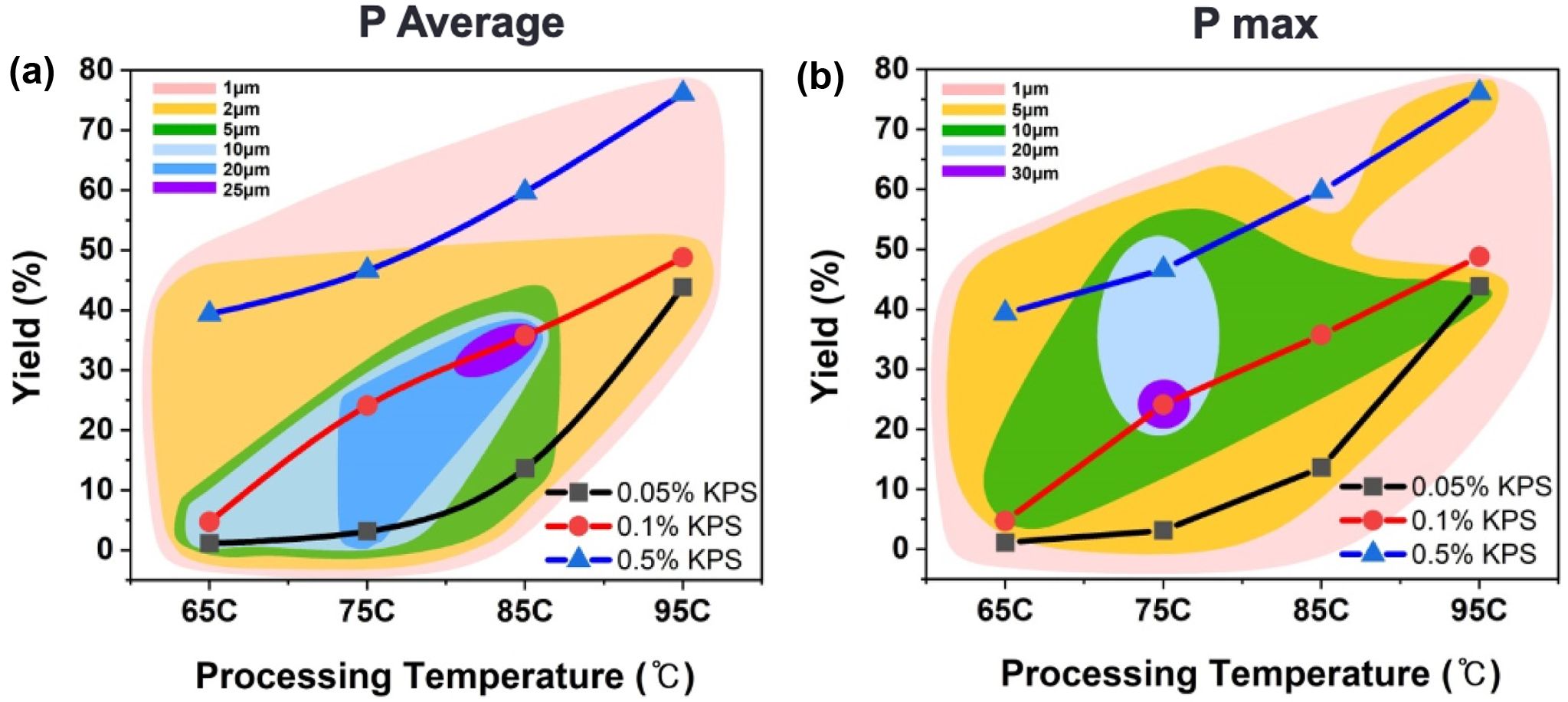
From the above finding the optimized topographic charts has been presented in Figure 4. The average particle size is increases with rising temperatures from 65 ℃ to 95 ℃, as shown in Figure 4(a). The most pronounced yield percentage is observed at higher KPS concentrations (0.5%). The maximum average particle size is 25 µm at 0.1% KPS concentration and 85 ℃ temperature. Correspondingly, Figure 4(b) shows the similar trends for maximum particle size, indicating a similar dependency on temperature and KPS concentration. It has been shown that, at lower temperatures (65 ℃), particles yield is consistently smaller across all KPS concentrations, while at higher temperatures, particularly 95 ℃, the particle yield is higher, predominantly at 0.5% KPS. In contrast to the particle size, specifically, at 75 ℃ temperature and 0.1% KPS concentration, the maximum particles size is 30 µm.
|
Figure 1 (a) By passage of each stage, the particles become larger by sticking together and this process results in discontinuous particle size growth; (b) schematic representation of nucleation, condensation and agglomeration. |

|
Figure 2 SEM images of PS particles synthesized under various temperatures and KPS concentrations. |
|
Figure 3 Size distribution diagrams of thermal conditions: (a) T = 65 ℃; (b) T = 75 ℃; (c) T = 85 ℃; (d) T = 95 ℃. |
|
Figure 4 Topographic chart of size distribution with all the conditions of temperature and initiator concentration: (a) average size of all particles; (b) the maximum size of the particles. |
|
Table 2 Statistical Representation of the Size of Synthesized PS Particles in Each Reaction Condition |
In this work a systematic synthesis method has been developed that allows the customization of particle sizes between 1-100 µm by thermodynamically manipulating nucleation, condensation, and agglomeration. In addition, significant control over the final particle size is achieved by modulating the collision frequency by manipulating key thermodynamic parameters such as temperature and initiator concentration. This approach not only achieves monodisperse particle synthesis, but also prioritizes the thermodynamic mechanism over agglomeration, enabling precise size control within the desired range of 1 to 100 µm. This dynamic growth process not only allows precise size distribution control but also opens up new possibilities for scalability, providing a promising avenue for applications where a broad range of particle sizes is crucial.
- 1. Girard, J.; Brunetto, P. S.; Braissant, O.; Rajacic, Z.; Khanna, N.; Landmann, R.; Daniels, A. U.; Fromm, K. M. Development of a Polystyrene Sulfonate/Silver Nanocomposite with Self-Healing Properties for Biomaterial Applications. Comptes. Rendu. Chim. 2013, 16, 550-556.
-

- 2. Sivaprasad, S.; Byju, S. K.; Prajith, C.; Shaju, J.; Rejeesh, C. R. Development of a Novel Mycelium Bio-Composite Material to Substitute for Polystyrene in Packaging Applications. Mater. Today. Proc. 2021, 47, 5038-5044.
-

- 3. Janajreh, I.; Adeyemi, I.; Elagroudy, S. Gasification Feasibility of Polyethylene, Polypropylene, Polystyrene Waste and Their Mixture: Experimental Studies and Modeling. Sust. Energ. Technol. Ass. 2020, 39, 100684.
-

- 4. Zhang, Y.; Yu, Z.; Yue, Z.; Gao, J.; Wu, S.; Zhang, Z.; Li, G. Rapid Determination of Trace Nitrofurantoin in Cosmetics by Surface Enhanced Raman Spectroscopy Using Nanoarrayed Hydroxyl Polystyrene‐based Substrate. J. Raman Spectrosc. 2019, 50, 1094-1102.
-

- 5. Kim, G.; Park, K.; Zheng, Z.; Choi, S.; Jin, S. Cross-Linker-Controlled Ostwald Ripening in Emulsion Polymerization of Hollow Copolymer Nanoparticles. J. Phys. Chem. B. 2020, 124, 10276-10281.
-

- 6. Lee, J.; Ha, J. U.; Choe, S.; Lee, C.-S.; Shim, S. E. Synthesis of Highly Monodisperse Polystyrene Microspheres via Dispersion Polymerization Using an Amphoteric Initiator. J. Colloid. Interface Sci. 2006, 298, 663-671.
-

- 7. Fernandes, L. S. L.; Cellet, T. S. P.; Souza, E. M. B. D.; Sayer, C.; Rubira, A. F.; Dariva, C.; Oliveira, J. A. Development of a System by Atomization for the Formation of Polymeric Particles in Micro and Sub-Micro Scales. Colloids Surf. A Physicochem. Eng. Asp. 2014, 451, 1-6.
-

- 8. Liu, B.; Zhang, M.; Liu, Y.; Tan, Z.; Zhou, C.; Zhang, H. Particle Nucleation and Growth in the Emulsion Polymerization of Styrene: Effect of Monomer/Water Ratio and Electrolyte Concentration. J. Macromol. Sci. Part A. 2015, 52, 147-154.
-

- 9. Karandikar, P.; Gupta, M. Synthesis of Functional Particles by Condensation and Polymerization of Monomer Droplets in Silicone Oils. Langmuir 2017, 33, 7701-7707.
-

- 10. Ashraf, M. A.; Peng, W.; Zare, Y.; Rhee, K. Y. Effects of Size and Aggregation/Agglomeration of Nanoparticles on the Interfacial/Interphase Properties and Tensile Strength of Polymer Nanocomposites. Nanoscale. Res. Lett. 2018, 13, 214.
-

- 11. Lee, W. H.; Booth, J. R.; Bon, S. A. F. On Particle Size Distributions in Catalytic Chain Transfer Emulsion Polymerization: Chain-Extension and the Use of Derived Macromonomers as Reactive Surfactants in Emulsion Polymerization. Biomacromolecules 2020, 21, 4599-4614.
-

- 12. Lim, H.-H.; Oh, Y.-S.; Shin, H.-S. Determination of N-Nitrosodimethylamine and N-Nitrosomethylethylamine in Drug Substances and Products of Sartans, Metformin and Ranitidine by Precipitation and Solid Phase Extraction and Gas Chromatography–Tandem Mass Spectrometry. J. Pharm. Biomed. Anal. 2020, 189, 113460.
-

- 13. De Vos, J.; Broeckhoven, K.; Eeltink, S. Advances in Ultrahigh-Pressure Liquid Chromatography Technology and System Design. Anal. Chem. 2016, 88, 262-278.
-

- 14. Gama, M. R.; Bottoli, C. B. G. Nanomaterials in Liquid Chromatography: Recent Advances in Stationary Phases. In Nanomaterials in Chromatography; Elsevier, 2018; pp 255-297.
-

- 15. Chern, C. S. Emulsion Polymerization Mechanisms and Kinetics. Prog. Polym. Sci. 2006, 31, 443-486.
- Polymer(Korea) 폴리머
- Frequency : Bimonthly(odd)
ISSN 2234-8077(Online)
Abbr. Polym. Korea - 2024 Impact Factor : 0.6
- Indexed in SCIE
 This Article
This Article
-
2026; 50(2): 295-302
Published online Mar 25, 2026
- 10.7317/pk.2026.50.2.295
- Received on Oct 1, 2025
- Revised on Jan 14, 2026
- Accepted on Jan 14, 2026
 Services
Services
- Full Text PDF
- Abstract
- ToC
- Acknowledgements
- Conflict of Interest
- Supporting Information
Introduction
Experimental
Result and Discussion
Conclusion
- References
Shared
 Correspondence to
Correspondence to
- Gunwoo Kim
-
Department of Organic Materials and Textile Engineering, Jeonbuk National University, 567 Baekje-daero, Deokjin-gu, Jeonju-si, Jeonbuk 54896, Korea
- E-mail: creaventure@jbnu.ac.kr
- ORCID:
0000-0002-4816-3664









 Copyright(c) The Polymer Society of Korea. All right reserved.
Copyright(c) The Polymer Society of Korea. All right reserved.